Analyzing Data
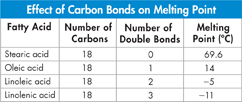
Comparing Fatty Acids
The table compares four different fatty acids. Although they all have the same number of carbon atoms, their properties vary.
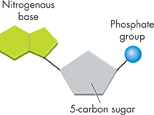
FIGURE 2–16 Nucleic Acids The monomers that make up a nucleic acid are nucleotides. Each nucleotide has a 5-carbon sugar, a phosphate group, and a nitrogenous base.
Nucleic Acids Nucleic acids are macromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus. Nucleic acids are polymers assembled from individual monomers known as nucleotides. Nucleotides consist of three parts: a 5-carbon sugar, a phosphate group (–PO4), and a nitrogenous base, as shown in Figure 2–16. Some nucleotides, including the compound known as adenosine triphosphate (ATP), play important roles in capturing and transferring chemical energy. Individual nucleotides can be joined by covalent bonds to form a polynucleotide, or nucleic acid.
 Nucleic acids store and transmit hereditary, or genetic, information. There are two kinds of nucleic acids: ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
Nucleic acids store and transmit hereditary, or genetic, information. There are two kinds of nucleic acids: ribonucleic acid (RNA) and deoxyribonucleic acid (DNA). Protein Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. Proteins are polymers of molecules called amino acids, shown in Figure 2–17. Amino acids are compounds with an amino group (–NH2) on one end and a carboxyl group (–COOH) on the other end. Covalent bonds called peptide bonds link amino acids together to form a polypeptide. A protein is a functional molecule built from one or more polypeptides.  Some proteins control the rate of reactions and regulate cell processes. Others form important cellular structures, while still others transport substances into or out of cells or help to fight disease.
Some proteins control the rate of reactions and regulate cell processes. Others form important cellular structures, while still others transport substances into or out of cells or help to fight disease.
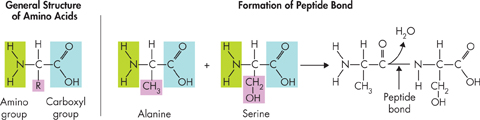
FIGURE 2–17 Amino Acids and Peptide Bonding Peptide bonds form between the amino group of one amino acid and the carboxyl group of another amino acid. A molecule of water (H2O) is released when the bond is formed. Note that it is the variable R-group section of the molecule that distinguishes one amino acid from another.
Table of Contents
- Formulas and Equations
- Applying Formulas and Equations
- Mean, Median, and Mode
- Estimation
- Using Measurements in Calculations
- Effects of Measurement Errors
- Accuracy
- Precision
- Comparing Accuracy and Precision
- Significant Figures
- Calculating With Significant Figures
- Scientific Notation
- Calculating With Scientific Notation
- Dimensional Analysis
- Applying Dimensional Analysis