2.4 Chemical Reactions and Enzymes
 What happens to chemical bonds during chemical reactions?
What happens to chemical bonds during chemical reactions? How do energy changes affect whether a chemical reaction will occur?
How do energy changes affect whether a chemical reaction will occur? What role do enzymes play in living things and what affects their function?
What role do enzymes play in living things and what affects their function?
chemical reaction • reactant • product • activation energy • catalyst • enzyme • substrate
Concept Map As you read, make a concept map that shows the relationship among the vocabulary terms in this lesson.
THINK ABOUT IT Living things, as you have seen, are made up of chemical compounds—some simple and some complex. But chemistry isn't just what life is made of—chemistry is also what life does. Everything that happens in an organism—its growth, its interaction with the environment, its reproduction, and even its movement—is based on chemical reactions.
Chemical Reactions
 What happens to chemical bonds during chemical reactions?
What happens to chemical bonds during chemical reactions?
A chemical reaction is a process that changes, or transforms, one set of chemicals into another. An important scientific principle is that mass and energy are conserved during chemical transformations. This is also true for chemical reactions that occur in living organisms. Some chemical reactions occur slowly, such as the combination of iron and oxygen to form an iron oxide called rust. Other reactions occur quickly. The elements or compounds that enter into a chemical reaction are known as reactants. The elements or compounds produced by a chemical reaction are known as products.  Chemical reactions involve changes in the chemical bonds that join atoms in compounds. An important chemical reaction in your bloodstream that enables carbon dioxide to be removed from the body is shown in Figure 2–19.
Chemical reactions involve changes in the chemical bonds that join atoms in compounds. An important chemical reaction in your bloodstream that enables carbon dioxide to be removed from the body is shown in Figure 2–19.
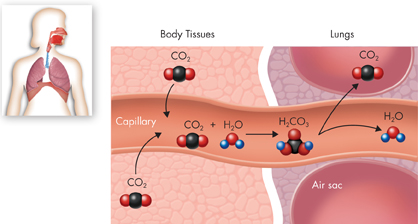
FIGURE 2–19 Carbon Dioxide in the Bloodstream As it enters the blood, carbon dioxide reacts with water to produce carbonic acid (H2CO3), which is highly soluble. This reaction enables the blood to carry carbon dioxide to the lungs. In the lungs, the reaction is reversed and produces carbon dioxide gas, which you exhale.
d
Table of Contents
- Formulas and Equations
- Applying Formulas and Equations
- Mean, Median, and Mode
- Estimation
- Using Measurements in Calculations
- Effects of Measurement Errors
- Accuracy
- Precision
- Comparing Accuracy and Precision
- Significant Figures
- Calculating With Significant Figures
- Scientific Notation
- Calculating With Scientific Notation
- Dimensional Analysis
- Applying Dimensional Analysis