▸ Structure and Function More than 20 different amino acids are found in nature. All amino acids are identical in the regions where they may be joined together by covalent bonds. This uniformity allows any amino acid to be joined to any other amino acid—by bonding an amino group to a carboxyl group. Proteins are among the most diverse macromolecules. The reason is that amino acids differ from each other in a side chain called the R-group, which have a range of different properties. Some R-groups are acidic and some are basic. Some are polar, some are nonpolar, and some even contain large ring structures.
▸ Levels of Organization Amino acids are assembled into polypeptide chains according to instructions coded in DNA. To help understand these large molecules, scientists describe proteins as having four levels of structure. A protein's primary structure is the sequence of its amino acids. Secondary structure is the folding or coiling of the polypeptide chain. Tertiary structure is the complete, three-dimensional arrangement of a polypeptide chain. Proteins with more than one chain are said to have a fourth level of structure, describing the way in which the different polypeptides are arranged with respect to each other. Figure 2–18 shows these four levels of structure in hemoglobin, a protein found in red blood cells that helps to transport oxygen in the bloodstream. The shape of a protein is maintained by a variety of forces, including ionic and covalent bonds, as well as van der Waals forces and hydrogen bonds. In the next lesson, you will learn why a protein's shape is so important.
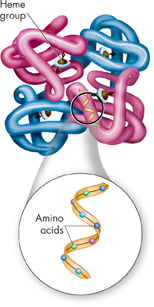
FIGURE 2–18 Protein Structure The protein hemoglobin consists of four subunits. The iron-containing heme group in the center of each subunit gives hemoglobin its red color. An oxygen molecule binds tightly to each heme molecule. Interpret Visuals How many levels of organization does hemoglobin have?
2.3 Assessment

-
Review What are the major elements of life?
Relate Cause and Effect What properties of carbon explain carbon's ability to form different large and complex structures?
-
Review Name four groups of organic compounds found in living things.
Explain Describe at least one function of each group of organic compound.
Infer Why are proteins considered polymers but lipids not?
VISUAL THINKING
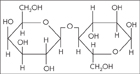
A structural formula shows how the atoms in a compound are arranged.
Observe What atoms constitute the compound above?
Classify What class of macromolecule does the compound belong to?

Table of Contents
- Formulas and Equations
- Applying Formulas and Equations
- Mean, Median, and Mode
- Estimation
- Using Measurements in Calculations
- Effects of Measurement Errors
- Accuracy
- Precision
- Comparing Accuracy and Precision
- Significant Figures
- Calculating With Significant Figures
- Scientific Notation
- Calculating With Scientific Notation
- Dimensional Analysis
- Applying Dimensional Analysis