4.3 Modern Atomic Theory
Reading Focus
Key Concepts
 What can happen to electrons when atoms gain or lose energy?
What can happen to electrons when atoms gain or lose energy? What model do scientists use to describe how electrons behave in atoms?
What model do scientists use to describe how electrons behave in atoms? What is the most stable configuration of electrons in an atom?
What is the most stable configuration of electrons in an atom?
Vocabulary
energy levels
electron cloud
orbital
electron configuration
ground state
Reading Strategy
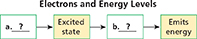
Sequencing Copy the flow chart. After you read, complete the description of how a gain or loss of energy affects atoms.
Have you ever wondered what produces the different colors in a fireworks display? Why does one explosion produce red light and another explosion produce green light? The people who make fireworks know that certain compounds will produce certain colors of light when they are heated. For example, compounds containing the element strontium produce red light when they are heated. Compounds containing barium produce green light.
You have seen two things that can happen when atoms absorb energy—an increase in kinetic energy or a phase change. But there is another possibility. The energy may be temporarily absorbed by the atom and then emitted as light. The colors in a fireworks display are a clue to how electrons are arranged in atoms.
Bohr's Model of the Atom
You may have seen diagrams of an atom that look like a solar system with planets revolving around a sun. These diagrams are based on a model of the atom that was developed by Niels Bohr (1885–1962), a Danish physicist who worked for a while with Rutherford. Bohr agreed with Rutherford's model of a nucleus surrounded by a large volume of space. But Bohr's model did something that Rutherford's model did not do. It focused on the electrons. A description of the arrangement of electrons in an atom is the centerpiece of the modern atomic model.
Figure 13 Fireworks are often displayed above the Lincoln Memorial in Washington, D.C. The red light was produced by a strontium compound.