CHAPTER 9 Study Guide
9.1 Carbon Compounds
 Key Concepts
Key Concepts
Diamond, graphite, and fullerenes are forms of carbon. Diamond is a network solid. Graphite forms layers that slide past one another. Fullerenes are large hollow spheres or cages of carbon.
Factors that determine the properties of a hydrocarbon are the number of carbon atoms and how the atoms are arranged. Hydrocarbons can form a straight chain, a branched chain, or a ring.
There are three types of unsaturated hydrocarbons—alkenes, alkynes, and aromatic hydrocarbons.
Three types of fossil fuels are coal, petroleum, and natural gas. The primary products of the complete combustion of fossil fuels are carbon dioxide and water.
Vocabulary
organic compound, p. 262;
network solid, p. 263;
hydrocarbon, p. 264;
saturated hydrocarbon, p. 264;
isomers, p. 265;
unsaturated hydrocarbon, p. 266;
aromatic hydrocarbons, p. 266;
fossil fuels, p. 267
9.2 Substituted Hydrocarbons
 Key Concepts
Key Concepts
The functional group in an alcohol is a hydroxyl group, −OH. The functional group in an organic acid is a carboxyl group, −COOH. The functional group in an amine is an amino group, −NH2.
Esters form when organic acids react with alcohols.
Vocabulary
9.3 Polymers
 Key Concepts
Key Concepts
Polymers can be classified as natural polymers or synthetic polymers.
Rubber, nylon, and polyethylene are three examples of compounds that can be synthesized.
Four types of polymers produced in plant and animal cells are starches, cellulose, nucleic acids, and proteins.
Vocabulary
polymer, p. 275;
monomers, p. 275;
carbohydrates, p. 278;
nucleic acids, p. 279;
amino acid, p. 280;
protein, p. 280
9.4 Reactions in Cells
 Key Concepts
Key Concepts
During photosynthesis, energy from sunlight is converted into chemical energy.
During cellular respiration, the energy stored in the products of photosynthesis is released.
Enzymes and vitamins help cells function efficiently at normal body temperature.
Vocabulary
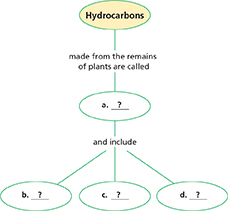
Thinking Visually
Concept Map Copy the concept map onto a sheet of paper. Use information from the chapter to complete it.