Do you know the smell of rotten fish? Then you've encountered a type of substituted hydrocarbon called an amine. Amines are organic bases.  The functional group in an amine is an amino group, −NH2. Amines are found in paints, dyes, and disinfectants. In Section 9.3 you will study the role organic bases play in the formation of organic compounds that are essential for life.
The functional group in an amine is an amino group, −NH2. Amines are found in paints, dyes, and disinfectants. In Section 9.3 you will study the role organic bases play in the formation of organic compounds that are essential for life.
Esters
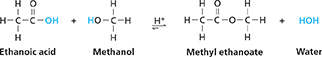
One group of substituted hydrocarbons accounts for the flavors of many foods and the pleasant odor of many flowers. These compounds are known as esters.  Esters form when organic acids react with alcohols. The second product of the reaction is water. For example, ethanoic acid can react with methanol to produce methyl ethanoate (methyl acetate). The reaction is reversible.
Esters form when organic acids react with alcohols. The second product of the reaction is water. For example, ethanoic acid can react with methanol to produce methyl ethanoate (methyl acetate). The reaction is reversible.
Esters are used in many processed foods to produce flavors such as strawberry, banana, and grape. Flowers like the roses in Figure 12 produce esters and other compounds with distinctive odors that attract insects for pollination. Sometimes, the compounds produced by the plant mimic compounds produced by the insect.
Figure 12 Many compounds in rose petals contribute to the fragrance of a rose. Some of these compounds are esters, which tend to have pleasant, sweet odors.

Section 9.2 Assessment
Reviewing Concepts
 What functional groups are found in alcohols, organic acids, and organic bases?
What functional groups are found in alcohols, organic acids, and organic bases? Which types of compounds can react to produce esters?
Which types of compounds can react to produce esters?What is a substituted hydrocarbon?
When a halocarbon reacts with a base, what products are produced?
What are two properties of organic acids?
Critical Thinking
Classifying An unknown compound has no noticeable odor. Explain why the compound is unlikely to be an organic acid, an organic base, or an ester.
Inferring What kind of organic compound gives a vitamin-C tablet its sour taste?
Applying Concepts Name one kind of substituted hydrocarbon you would expect to find in artificially flavored grape jelly.
Connecting Concepts
Types of Reactions Alcohols can be made by reacting halocarbons with bases, or alkenes with water. Review the general types of reactions presented in Section 7.2. Which types best describe the two methods for producing an alcohol? Explain your choices.




