Measuring Heat Changes
A calorimeter is an instrument used to measure changes in thermal energy.  A calorimeter uses the principle that heat flows from a hotter object to a colder object until both reach the same temperature. According to the law of conservation of energy, the thermal energy released by a test sample is equal to the thermal energy absorbed by its surroundings. The calorimeter is sealed to prevent thermal energy from escaping.
A calorimeter uses the principle that heat flows from a hotter object to a colder object until both reach the same temperature. According to the law of conservation of energy, the thermal energy released by a test sample is equal to the thermal energy absorbed by its surroundings. The calorimeter is sealed to prevent thermal energy from escaping.
Figure 4 shows how a calorimeter can be used to measure the specific heat of aluminum. A known mass of water is added to the calorimeter. The mass of the sample of aluminum is measured. The aluminum is heated and then placed in the water. The calorimeter is sealed. As the aluminum cools off, the water is stirred to distribute thermal energy evenly. The water heats up until both the aluminum and the water are at the same temperature. The change in temperature of the water is measured. The thermal energy absorbed by the water is calculated using the specific heat equation. Since this same amount of thermal energy was given off by the sample of aluminum, the specific heat of aluminum can be calculated.
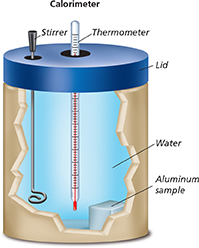
Figure 4 A calorimeter is used to measure specific heat. A sample to be tested is heated and placed in the calorimeter. The lid is put on and the temperature change is observed. Hypothesizing Why does the calorimeter need a stirrer?
Section 16.1 Assessment
Reviewing Concepts
 In what direction does heat flow on its own spontaneously?
In what direction does heat flow on its own spontaneously? How is the temperature of an object related to the average kinetic energy of its particles?
How is the temperature of an object related to the average kinetic energy of its particles? Name two variables that affect the thermal energy of an object.
Name two variables that affect the thermal energy of an object. What causes thermal expansion of an object when it is heated?
What causes thermal expansion of an object when it is heated? How do the temperature increases of different materials depend on their specific heats?
How do the temperature increases of different materials depend on their specific heats? What principle explains how a calorimeter is used to measure the specific heat of a sample material?
What principle explains how a calorimeter is used to measure the specific heat of a sample material?
Critical Thinking
Applying Concepts Why is it necessary to have regularly spaced gaps between sections of a concrete sidewalk?
Predicting An iron spoon and silver spoon have the same mass. Which becomes hotter when both are left in hot tea for one minute? (Hint: Use the specific heats given in Figure 3.)
Calculating If it takes 80.0 joules to raise the temperature of a material by 10.0°C, how much heat must be added to cause an additional increase of 20.0°C?
Math Practice
The specific heat of copper is 0.39 J/g•°C. How much heat is needed to raise the temperature of 1000.0 g of copper from 25.0°C to 45.0°C?
A peanut burned in a calorimeter transfers 18,200 joules to 100.0 g of water. What is the rise in the water's temperature? (Hint: Rearrange the specific heat formula to solve for ΔT.)




