CHAPTER 7 Assessment
Interactive Textbook with assessment at PHSchool.com

Reviewing Content
Choose the letter that best answers the question or completes the statement.
In the following equation, what are the reactants?
NaHCO3 and HCl
NaHCO3 and NaCl
HCl and NaCl
NaCl, H2O, and CO2
Which of the following is a statement of the law of conservation of mass?
Mass is created but not destroyed.
Mass is destroyed but not created.
Mass is neither created nor destroyed.
Mass is both created and destroyed, depending on the chemical reaction.
The mass of a hydrogen atom is 1.0 amu, and the mass of a carbon atom is 12.0 amu. What is the molar mass of methane, CH4?
13.0 amu
13.0 g
16.0 amu
16.0 g
In what type of reaction does one reactant form two or more products?
synthesis
decomposition
single replacement
double replacement
What particle is transferred from one atom to another in a redox reaction?
electron
neutron
proton
nucleus
Which of the following is a single replacement?
How are reactions related to chemical bonds?
Bonds in the reactants are broken, and bonds in the products are formed.
Bonds in the products are broken, and bonds in the reactants are formed.
Bonds in both the reactants and products are broken.
Bonds are formed in both the reactants and the products.
What type of reaction always releases energy?
endothemic
exothermic
decomposition
oxidation-reduction
In general, an increase in temperature
increases reaction rate.
decreases reaction rate.
does not affect reaction rate.
acts as a catalyst.
What takes place at chemical equilibrium?
Reactants form more quickly than products.
Products form more quickly than reactants.
Reactants and products stop forming.
Reactants and products form at the same rate.
Understanding Concepts
Write the following chemical equation in words.
Explain how a balanced chemical equation shows that mass is conserved.
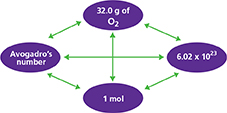
Explain the following diagram in your own words.
Compare oxidation and reduction.
Paper burns by combining with oxygen. Why doesn't paper burn every time it contacts oxygen?
Give an example of a chemical reaction that occurs slowly. Give another example of a chemical reaction that occurs quickly.
Answer Questions 17–18 based on the equation below.
How would you adjust the temperature to increase the amount of product?
Does the removal of hydrogen gas as it is produced shift the reaction to the left or the right?