The Combined Gas Law
The relationships described by Boyle's law and Charles's law can be described by a single law. The combined gas law describes the relationship among the temperature, volume, and pressure of a gas when the number of particles is constant.
Combined Gas Law
The combined gas law is used to solve many problems involving gases.
Math Skills
The Combined Gas Law
A cylinder that contains air at a pressure of 100 kPa has a volume of 0.75 L. The pressure is increased to 300 kPa. The temperature does not change. Find the new volume of air.
Read and Understand
What information are you given?
Plan and Solve
What unknown are you trying to calculate?
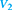
What expression can you use?
Cancel out the variable that does not change and rearrange the expression to solve for V2.
Replace each variable with its known value.
Look Back and Check
Is your answer reasonable?
Volume should decrease as pressure increases. The pressure tripled from 100 kPa to 300 kPa. The answer, 0.25 L, is one third the original volume, 0.75 L.
Math Practice
A gas has a volume of 5.0 L at a pressure of 50 kPa. What happens to the volume when the pressure is increased to 125 kPa? The temperature does not change.
Gas stored in a tank at 273 K has a pressure of 388 kPa. The safe limit for the pressure is 825 kPa. At what temperature will the gas reach this pressure?
At 10°C, the gas in a cylinder has a volume of 0.250 L. The gas is allowed to expand to 0.285 L. What must the final temperature be for the pressure to remain constant? (Hint: Convert from degrees Celsius to kelvins using the expression )




