Fluorescent Light
In a process called fluorescence (floo uh res uns), a material absorbs light at one wavelength and then emits light at a longer wavelength. A phosphor is a solid material that can emit light by fluorescence.  Fluorescent light bulbs emit light by causing a phosphor to steadily emit photons. A fluorescent bulb, such as the one in Figure 28, is a glass tube that contains mercury vapor. Inside, the glass is coated with phosphors.
Fluorescent light bulbs emit light by causing a phosphor to steadily emit photons. A fluorescent bulb, such as the one in Figure 28, is a glass tube that contains mercury vapor. Inside, the glass is coated with phosphors.
Figure 28 The electrodes in a fluorescent bulb emit electrons that cause the mercury atoms to emit ultraviolet rays. These rays cause the phosphor coating to emit light.
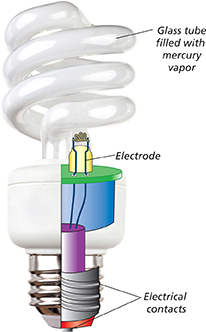
When electric current flows through a fluorescent bulb, small pieces of metal called electrodes heat up and emit electrons. The electrons hit atoms of the mercury vapor, causing the mercury atoms to emit ultraviolet rays. The ultraviolet rays strike the phosphor coating on the inside of the tube and the atoms emit visible light.
You may have noticed that office buildings and schools use mostly fluorescent lights. Fluorescent tubes do not get as hot as incandescent bulbs because they emit most of their energy as light. This means that they use energy very efficiently. One 18-watt fluorescent tube provides the same amount of light as a 75-watt incandescent bulb, and the fluorescent tube lasts ten times longer.
What happens during fluorescence?
Quick Lab
Comparing Fluorescent and Incandescent Light
Materials
spectroscope, clear incandescent bulb, fluorescent bulb, colored pencils
Procedure 
Turn on a clear, incandescent bulb.
CAUTION Incandescent bulbs get quite hot after they have been on for some time. Observe the spectrum of the light coming from the incandescent bulb through a spectroscope.
Use colored pencils to draw this spectrum. Label the source of the spectrum.
Repeat Steps 1 and 2 with a fluorescent bulb.
Analyze and Conclude
Comparing and Contrasting How do the spectra produced by incandescent and fluorescent lights compare?
Drawing Conclusions During fluorescence, electrons absorb energy and move to specific higher energy levels. As they move back to a lower energy level, they release energy in the form of light. How does this fact help explain the appearance of the spectrum of fluorescent light?





