Proteins
Recall that organic acids contain a −COOH group and organic bases, or amines, contain an −NH2 group. There is one substituted hydrocarbon that contains both groups. An amino acid is a compound that contains both carboxyl and amino functional groups in the same molecule. There are about 20 amino acids that your body needs to function.
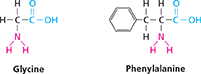
Your cells can manufacture some, but not all, of the amino acids. For example, your body can make glycine, but not phenylalanine. The essential amino acids that your body cannot make must come from foods like those in Figure 17.
Figure 17 The foods shown are all good protein sources. Classifying Group the foods into three or four categories.

Your cells use amino acids as the monomers for constructing protein polymers. A protein is a polymer in which at least 100 amino acid monomers are linked through bonds between an amino group and a carboxyl group. The instructions for making proteins are stored in DNA. Proteins make up the fibers of your muscles, your hair and fingernails, and the hemoglobin in your blood. Your body may contain as many as 300,000 different proteins.
Section 9.3 Assessment
Reviewing Concepts
 Describe a way that polymers can be classified.
Describe a way that polymers can be classified. Name three synthetic polymers.
Name three synthetic polymers. What are four types of polymers that can be found in the cells of organisms?
What are four types of polymers that can be found in the cells of organisms?What are the three parts of a nucleotide?
What holds the bases together in DNA?
What two functional groups are found in amino acids?
Critical Thinking
Using Analogies Which natural polymers are like a train with identical freight cars and which are like a train with a mixture of different cars?
Inferring There is another system for classifying carbohydrates that uses the categories monosaccharides, disaccharides, and polysaccharides. Where would you place glucose, sucrose, and cellulose in this classification system? Give a reason for your answer.
Writing in Science
Compare-Contrast Paragraph Write a paragraph comparing natural and synthetic polymers in general. How are they similar? How are they different?




