Particle Accelerators
In Rutherford's transmutation experiment, the radioactive element radium was used as a source of alpha particles. However, sometimes transmutations will not occur unless the bombarding particles are moving at extremely high speeds. In order to perform such transmutations, scientists use devices called particle accelerators. In a particle accelerator, charged particles can be accelerated to speeds very close to the speed of light. The fast-moving particles are guided toward a target, where they collide with atomic nuclei. With the help of particle accelerators, scientists have produced more than 3000 different isotopes.
Scientists also conduct collision experiments in order to study nuclear structure. Since the proton, neutron, and electron were discovered, more than 200 different subatomic particles have been detected. According to the current model of the atom, protons and neutrons are made up of even smaller particles called quarks. A quark is a subatomic particle theorized to be among the basic units of matter. Both protons and neutrons belong to a class of particles that are made up of three quarks. Six types of quarks are currently thought to exist. Two of these types were discovered at Fermi National Accelerator Laboratory, also known as Fermilab. Figure 14 shows one of the devices used at Fermilab to detect subatomic particles.
Figure 14 This particle detector records subatomic particles produced in the Tevatron, the most powerful particle accelerator in the United States. The Tevatron is located at Fermilab in Batavia, Illinois.

Section 10.3 Assessment
Reviewing Concepts
 How do scientists perform artificial transmutations?
How do scientists perform artificial transmutations? How are transuranium elements produced?
How are transuranium elements produced?How does artificial transmutation differ from nuclear decay?
Write the equation for the transmutation that occurs when an alpha particle combines with an oxygen-16 atom, emitting a proton.
Does fermium-257 undergo nuclear decay? Explain.
Critical Thinking
Predicting Bombarding a lithium-6 atom with a neutron produces helium-4 and another particle. What is that particle?
Predicting Curium was first synthesized by bombarding a target isotope with alpha particles, which produced curium-242 and a neutron. What was the target isotope? (Hint: Use the symbol
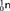 to represent a neutron.)
to represent a neutron.)Inferring Why can't the transuranium elements be made by exposing other elements to naturally occurring alpha radiation?
Writing in Science
Summary Write a brief summary of the first artificial transmutation, performed by Ernest Rutherford. (Hint: Your summary should describe an example of a nuclear reaction.)




