2.1 Classifying Matter
Reading Focus
Key Concepts
 Why are elements and compounds classified as pure substances?
Why are elements and compounds classified as pure substances? How do mixtures differ from pure substances?
How do mixtures differ from pure substances? What is the main difference among solutions, suspensions, and colloids?
What is the main difference among solutions, suspensions, and colloids?
Vocabulary
pure substance
element
atom
compound
heterogeneous mixture
homogeneous mixture
solution
suspension
colloid
Reading Strategy
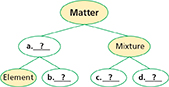
Summarizing Copy the diagram below. As you read, complete the classification of matter.
Each piece of clothing sold has a care label, which lists recommended cleaning methods for the clothing. For a sweater, the instructions might say to machine wash the sweater using a gentle cycle, and then tumble dry at a low temperature. They might say to hand wash the sweater in cold water and lay the sweater flat to dry. The label might even say, “Dry clean only.”
Why is it necessary to put care instructions on a label? The same cleaning method will not work for all materials. For example, a shirt made from 100 percent cotton may need to be ironed after washing. But a shirt made from a cotton and polyester blend may come out of the dryer wrinkle free. A wool jacket often needs to be dry cleaned because wool can shrink when washed in water.
The tendency to wrinkle when washed is a property of cotton. The tendency not to wrinkle when washed is a property of polyester. The tendency to shrink when washed is a property of wool. Cotton, wool, and polyester have different properties because they have different compositions. The word composition comes from a Latin word meaning “a putting together,” or the combining of parts into a whole. Based on their compositions, materials can be divided into pure substances and mixtures.
Figure 1 You can use the care labels on clothing to sort laundry into batches for cleaning. The care label shown is for a wool sweater that needs to be dry cleaned or washed by hand.