The heat (Q) absorbed by a material equals the product of the mass (m), the specific heat (c), and the change in temperature (▲T).
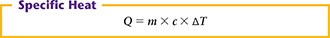
In this formula, heat is in joules, mass is in grams, specific heat is in J/g•°C, and the temperature change is in degrees Celsius.
Math Skills
Calculating Specific Heat
An iron skillet has a mass of 500.0 grams. The specific heat of iron is 0.449 J/g•°C. How much heat must be absorbed to raise the skillet's temperature by 95.0°C?
Read and Understand
What information are you given?
Plan and Solve
What unknown are you trying to calculate?
Amount of heat needed, Q = ?
What formula contains the given quantities and the unknown?
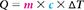
Replace each variable with its known value.
Look Back and Check
Is your answer reasonable?
Round off the data to give a quick estimate.
This is close to 21.4 kJ, so the answer is reasonable.
Math Practice
How much heat is needed to raise the temperature of 100.0 g of water by 85.0°C?
How much heat is absorbed by a 750-g iron skillet when its temperature rises from 25°C to 125°C?
In setting up an aquarium, the heater transfers 1200 kJ of heat to 75,000 g of water. What is the increase in the water's temperature? (Hint: Rearrange the specific heat formula to solve for ?▲.)
To release a diamond from its setting, a jeweler heats a 10.0-g silver ring by adding 23.5 J of heat. How much does the temperature of the silver increase?
What mass of water will change its temperature by 3.0°C when 525 J of heat is added to it?





