Data Analysis
Exploring Boiling Points of Chlorocarbons
Use the data given to construct a graph with Number of Chlorine Atoms, from 0 to 4, on the horizontal axis and Boiling Point on the vertical axis. Draw a smooth curve connecting the points.
Using Graphs Predict the boiling point of dichloromethane.
Relating Cause and Effect What effect does increasing the number of chlorine atoms have on the boiling point?
Boiling Points of Methane and Its Substituted Chlorocarbons
Name
Formula
Boiling Point (°C)
Methane
CH4
−161
Chloromethane
CH3Cl
−24
Dichloromethane
CH2Cl2

Trichloromethane
CHCl3
61
Tetrachloromethane
CCl4
77
Formulating Hypotheses Why is the boiling point of CH3Br higher than the boiling point of CH3Cl? (Hint: Compare the atomic masses for bromine and chlorine.)
Alcohols
Methanol, CH3OH, is used as a fuel in some motorcycles. Ethanol, CH3CH2OH, is often mixed with gasoline to help the gasoline burn more completely. Methanol and ethanol are alcohols. The name of an alcohol ends in −ol.  The functional group in an alcohol is a hydroxyl group, −OH. When a halocarbon reacts with a base, the products are an alcohol and salt.
The functional group in an alcohol is a hydroxyl group, −OH. When a halocarbon reacts with a base, the products are an alcohol and salt.
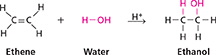
An alcohol can also be made by reacting an alkene with water.
Organic Acids and Bases
The sharp, sour taste of a lemon comes from citric acid, an organic acid.  The functional group in organic acids is a carboxyl group, −COOH. Names of organic acids end in -oic. Organic acids tend to have sharp tastes and strong odors. The simplest organic acid is methanoic acid, which is also known as formic acid. Vinegar is a solution of water and the organic acid ethanoic acid, which is usually referred to as acetic acid. If the ants in Figure 11 sprayed the formic acid they produce on your skin, your skin would itch or burn.
The functional group in organic acids is a carboxyl group, −COOH. Names of organic acids end in -oic. Organic acids tend to have sharp tastes and strong odors. The simplest organic acid is methanoic acid, which is also known as formic acid. Vinegar is a solution of water and the organic acid ethanoic acid, which is usually referred to as acetic acid. If the ants in Figure 11 sprayed the formic acid they produce on your skin, your skin would itch or burn.

Figure 11 Birds sometimes will sit on an ants' nest and allow the ants to crawl over them. The methanoic acid sprayed by the ants is a natural pesticide that kills mites in the birds' feathers.





