Forensics Lab

Using Flame Tests
Forensic scientists use various approaches to distinguish different substances. In this lab, you will observe the flame colors of several substances and use the data to determine the identity of an unknown substance.
Problem How can the color of a flame be used to distinguish substances?
Materials
solutions of calcium chloride, boric acid, potassium chloride, copper(II) sulfate, sodium chloride, and an unknown
Bunsen burner
nichrome wire loop
dilute solution of hydrochloric acid
wash bottle with distilled water
Skills Observing, Predicting, Using Data Tables
Procedure 
Part A: Observing Flame Colors
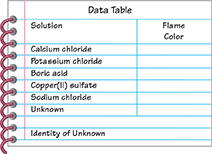
Make a copy of the data table shown.
Light the Bunsen burner. CAUTION Put on safety goggles and a lab apron. Tie back loose hair and clothing before working with a flame.
Dip the wire loop into the calcium chloride solution and then place the loop in the flame as shown. Observe and record the color of the flame.
Clean the loop by dipping it into hydrochloric acid. Then, while holding the loop over a sink, rinse away the acid with distilled water. CAUTION Keep hydrochloric acid away from your skin and clothing. Do not breathe in its vapor.
Repeat Steps 3 and 4 with each of the other solutions. Be careful not to transfer any solution from one container to another. CAUTION These chemicals are poisonous. Do not let them get on your skin.
Part B: Examining an Unknown Solution
Obtain the unknown solution from your teacher.
Repeat Steps 3 and 4 using the unknown solution. Compare your observations with the other data you recorded to identify the unknown. CAUTION Wash your hands thoroughly before leaving the laboratory.
Analyze and Conclude
Comparing and Contrasting Is there a relationship between the color of the flame and the color of the solution?
Formulating Hypotheses How do these substances produce light of different colors?
Drawing Conclusions A forensic scientist does a flame test on a substance that was found at a crime scene. What might the scientist conclude if the flame turns green?
Go Further
There is another test that you can use to distinguish elements by color. With your teacher supervising, dip a wire loop in borax. Heat the loop in a flame until the borax melts. Remove the loop from the flame and let the borax cool. It will form a clear glass bead. Dip the bead in a tiny sample of solid copper sulfate and return the loop to the flame for a few seconds. Remove the loop and observe the color of the bead as it cools.