CONCEPTS in Action
Chipping In
Tasks done by computers that filled an entire room in the 1950s are now done by devices the size of a credit card. This miniaturization in the electronics industry is due to semiconductors.
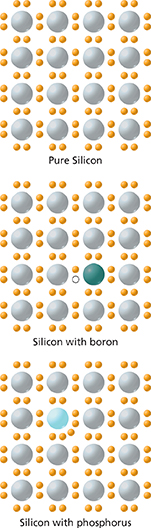
Semiconductors are solid substances, such as silicon, that have poor electrical conductivity at ordinary temperatures. Silicon has four valence electrons. In pure silicon, each atom forms single bonds with four other atoms. This arrangement leaves no electrons free to move through the silicon. The conductivity of silicon is greatly improved by adding small amounts of other elements to silicon, a process called doping.
Doping
An element with five valence electrons, such as phosphorus, can be added to silicon. After a phosphorus atom bonds with four atoms, there is an extra electron that is free to move. Silicon doped with phosphorus is called n-type silicon because electrons have a negative charge. An element with three valence electrons, such as boron, can be added to silicon. Adding boron leaves holes to which electrons can move from neighboring atoms. Because the lack of an electron has the effect of a positive charge, silicon with boron is called p-type silicon.
Wafers and chips
Silicon crystals are cut into wafer-thin slices. Each wafer can be made into hundreds of silicon chips.