 How It Works
How It Works
Automobile Safety: Air Bags
Air bags are inflatable cushions built into a car's steering wheel or dashboard. In a crash, the bags inflate, protecting both the driver and the passenger. The whole process takes 0.04 second. Interpreting Diagrams What is the source of the gas that fills an air bag?

Testing air bags
Air bags have been shown to reduce the risk of serious injury in a head-on collision by 30 percent. New cars have air bags on both the driver and passenger sides.
Collision A collision triggers the car's crash sensors, which send an electrical signal to the igniter in the steering wheel.
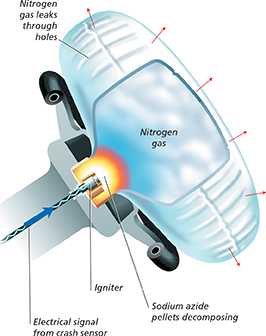
Air bag inflates The igniter sets off a combustion reaction that heats up the sodium azide (NaN3) contained in the ignition unit. When it is heated, the sodium azide decomposes into metallic sodium (Na) and nitrogen gas (N2). The nitrogen gas inflates the air bag. The decomposition reaction is 2NaN3 → 2Na → 3N2.

Air bag deflates The nitrogen escapes through tiny holes in the bag, causing immediate deflation of the air bag. Because sodium is dangerous (due to its high reactivity), the ignition unit also contains other chemicals that react with sodium to form a nontoxic material.





