 Standardized Test Prep
Standardized Test Prep
Test-Taking Tip
Make Logical Connections
A cause-and-effect statement may seem to be true when it is false. The statement may seem true because the descriptions of the cause and the effect are both accurate. However, there is no logical connection between the cause and the effect. In the question below, the opening phrase contains an accurate statement about body temperature. Most of the answers are accurate, too. But only one answer provides a logical effect of the statement in the opening phrase.
Because body temperature cannot vary more than a few degrees from 37°C,
cellular respiration releases energy stored in covalent bonds.
carbohydrates are good sources of energy.
enzymes are required for reactions in cells.
fats are good sources of energy.
energy cannot be released in cells.
(Answer: C)
Choose the letter that best answers the question or completes the statement.
Benzene is classified as an aromatic hydrocarbon because
it has a strong odor.
it contains only carbon and hydrogen.
it is a compound found in coal.
it has an unsaturated ring structure.
it has a saturated ring structure.
How are photosynthesis and cellular respiration related?
They both occur in all living organisms.
They both consume more energy than they produce.
They both produce water and carbon dioxide.
Cellular respiration stores energy, and photosynthesis releases it.
Photosynthesis stores energy, and cellular respiration releases it.
Which type of compound forms when amino acids are linked together?
nucleic acid
protein
carbohydrate
organic acid
ester
What type of substituted hydrocarbon contains the functional group −OH?
halocarbon
alcohol
ester
organic base
organic acid
Use the diagram to answer Questions 5 and 6. The diagram shows a combustion apparatus used to identify hydrocarbons.
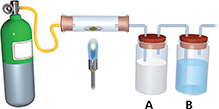
A sample of a hydrocarbon is in the glass tube.
What gas is in the green tank?
nitrogen
carbon dioxide
oxygen
methane
acetylene
The containers labeled A and B absorb the primary products of the reaction that occurs in the tube. Those products are
carbon dioxide and water.
carbon monoxide and water.
oxides of sulfur and nitrogen.
sulfuric and nitric acid.
carbon dioxide and carbon monoxide.




