Evidence Supporting Mendeleev's Table
Mendeleev named missing elements after elements in the same group. He gave the name eka-aluminum to the element that belonged one space below aluminum on the table. (Eka is a Sanskrit word meaning “one.”) Mendeleev predicted that eka-aluminum would be a soft metal with a low melting point and a density of 5.9 g/cm3.
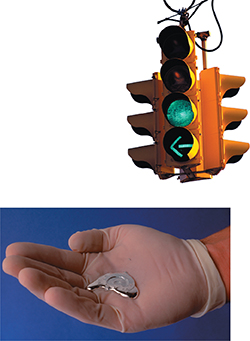
In 1875, a French chemist discovered a new element. He named the element gallium (Ga) in honor of France. (The Latin name for France is Gallia.) Gallium is a soft metal with a melting point of 29.7°C and a density of5.91 g/cm3. Figure 4 shows a sample of gallium and atraffic signal that uses gallium compounds.
The properties of gallium are remarkably similar to the predicted properties of eka-aluminum. Scientists concluded that gallium and eka-aluminum are the same element.  The close match between Mendeleev's predictions and the actual properties of new elements showed how useful his periodic table could be. The discovery of scandium (Sc) in 1879 and the discovery of germanium (Ge) in 1886 provided more evidence. With the periodic table, chemists could do more than predict the properties of new elements. They could explain the chemical behavior of different groups of elements.
The close match between Mendeleev's predictions and the actual properties of new elements showed how useful his periodic table could be. The discovery of scandium (Sc) in 1879 and the discovery of germanium (Ge) in 1886 provided more evidence. With the periodic table, chemists could do more than predict the properties of new elements. They could explain the chemical behavior of different groups of elements.
Figure 4 Gallium was discovered in 1875. Heat from a person's hand can melt gallium. In some traffic signals, there are tiny light emitting diodes (LEDs) that contain a compound of gallium. Comparing and Contrasting How does the melting point of gallium (29.7°C) compare to room temperature (about 25°C)?
Section 5.1 Assessment
Reviewing Concepts
 Describe how Mendeleev organized the elements into rows and columns in his periodic table.
Describe how Mendeleev organized the elements into rows and columns in his periodic table. How did the discovery of new elements such as gallium demonstrate the usefulness of Mendeleev's table?
How did the discovery of new elements such as gallium demonstrate the usefulness of Mendeleev's table?Scientists before Mendeleev had proposed ways to organize the elements. Why were Mendeleev's efforts more successful?
What characteristic of solitaire did Mendeleev use as a model for his periodic table?
Why did Mendeleev leave spaces in his table?
In general, how can a scientist test the correctness of a scientific model?
Critical Thinking
Inferring Explain why it would not have been possible for a scientist in 1750 to develop a table like Mendeleev's.
Predicting How was Mendeleev able to predict the properties of elements that had not yet been discovered?
Writing in Science
Writing to Persuade Write a paragraph about Mendeleev's periodic table. Use the paragraph to convince a reader that the periodic table is extremely useful to scientists. (Hint: Use specific facts to support your argument.)




