Exploration Lab
Predicting the Density of an Element
Density is a useful property for identifying and classifying elements. In this lab, you will determine the densities of three elements in Group 4A—silicon, tin, and lead. Then you will use your data to predict the density of another element in Group 4A—germanium.

Problem Can the densities of elements within a group be used to help predict the density of another element in the group?
Materials
unlined white paper
scissors
metric ruler
balance
forceps
silicon
tin
lead shot
50-mL graduated cylinder
graph paper
periodic table
 For the probeware version of this lab, see the Probeware Lab Manual, Lab 2.
For the probeware version of this lab, see the Probeware Lab Manual, Lab 2.
Skills Measuring, Observing, Using Graphs, Calculating
Procedure 
Part A: Measuring Mass
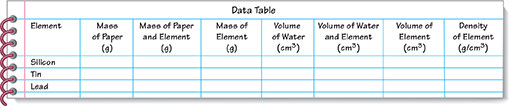
On a sheet of paper, make a copy of the data table shown.
Cut out three pieces of paper from a sheet of unlined white paper. Label one piece of paper Silicon, the second Tin, and the third Lead. Find the mass of each piece of paper and record it in your data table.
Using forceps, place the silicon onto the paper labeled Silicon, as shown. Find the mass of the silicon and the paper. Record this mass in your data table. Then, subtract the mass of the paper from the mass of the silicon and paper. Record the mass of silicon in your data table. Set the paper containing the silicon aside for now.
Repeat Step 3 to find the masses of tin and lead.
Part B: Measuring Volume
Place 25 mL of water in the graduated cylinder. Measure the volume of the water to the nearest 0.1 mL. Record the volume (in cm3) in your data table. (Hint:1 mL = 1 cm3)
Tilt the graduated cylinder and carefully pour the silicon from the paper into the graduated cylinder, as shown on page 151. Make sure that the silicon is completely covered by the water. Measure and record the volume of the water and silicon in your data table. Then, subtract the volume of water from the volume of the water and silicon. Record the result in your data table.
Repeat Steps 5 and 6 to find the volumes of tin and lead.