Standardized Test Prep
Test-Taking Tip
Calculating
When balancing chemical equations, follow these tips:
Never change the subscript in any formula. Change only the coefficients.
First, balance the atom that is most obviously unbalanced by placing a coefficient in front of the appropriate formula.
Next, balance other atoms by placing coefficients in front of appropriate formulas.
Finally, balance any atom that appears by itself in a formula (such as O2, H2, or Ca).
Count the number of atoms of each element on both sides of the equation. If they are not equal, continue balancing.
Practice using these tips in Questions 1 and 3.
Choose the letter that best answers the question or completes the statement.
Balance this chemical equation.
Questions 2 and 3 refer to the following reaction.
What type of reaction is described by this equation?
synthesis
decomposition
combustion
double replacement
single replacement
Balance the equation for the reaction between butane (C4H10) and oxygen, and then determine how many grams of CO2 are formed when
1.65 grams
3.79 grams
15.1 grams
20.0 grams
26.4 grams
Which equation describes a synthesis?
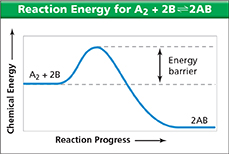
Use the energy diagram to answer Question 5.
What effect would a catalyst have on the reaction
It gives the products more energy.
It makes the reaction endothermic.
It gives the reacting particles more energy.
It provides a new reaction pathway for the reactants.
none of the above
The following equation shows the formation of ammonia (NH3) from nitrogen (N2) and hydrogen (H2). This reaction is in equilibrium.
If the pressure of the system is decreased, the equilibrium will shift
to the right because there are fewer moles of gas on the product side of the equation.
to the left because there are more moles of gas on the reactant side of the equation.
to the right because a drop in pressure brings the reactants together so they can react.
to the left because a drop in pressure pulls the product molecules apart.
in neither direction because pressure does not affect chemical equilibriums.