CHAPTER 8 Study Guide
8.1 Formation of Solutions
 Key Concepts
Key Concepts
Substances can dissolve in water in three ways—through dissociation, dispersion, and ionization.
Three physical properties of a solution that can differ from those of its solute and solvent are conductivity, freezing point, and boiling point.
During the formation of a solution, energy is either released or absorbed.
Factors that affect the rate of dissolving include surface area, temperature, and stirring.
Vocabulary
solute, p. 229; solvent, p. 229; dissociation, p. 229; dispersion, p. 230; ionization, p. 230
8.2 Solubility and Concentration
 Key Concepts
Key Concepts
Solutions are described as unsaturated, saturated, or supersaturated, depending on the amount of solute in solution.
Three factors that affect the solubility of a solute are the type of bonding in the solute and solvent, temperature, and pressure.
Concentration can be expressed as percent by volume, percent by mass, and molarity.

Vocabulary
solubility, p. 235; saturated solution, p. 236; unsaturated solution, p. 236; supersaturated solution, p. 236; concentration, p. 238; molarity, p. 239
8.3 Properties of Acids and Bases
 Key Concepts
Key Concepts
Acids taste sour, react with certain metals, and turn blue litmus paper red. Bases taste bitter, feel slippery, and turn red litmus paper blue.
The neutralization reaction between an acid and a base produces a salt and water.
Acids can be defined as proton donors, and bases can be defined as proton acceptors.
Vocabulary
acid, p. 240; indicator, p. 241; base, p. 242;
neutralization, p. 244; salt, p. 244
8.4 Strength of Acids and Bases
 Key Concepts
Key Concepts
The lower the pH value, the greater the H3O+ ion concentration is. The higher the pH value, the lower the H3O+ ion concentration is.
The strength of an acid or a base depends on the degree to which it dissociates or ionizes in water.
Strong acids and bases are good electrolytes because they produce many ions in solution.
Vocabulary
pH, p. 247; buffer, p. 248; electrolyte, p. 249
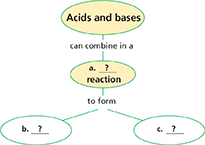
Thinking Visually
Concept Map Use the information on acids and bases to complete the concept map below.